Space Sickness
Space (“motion”) sickness was an expected complication of space flight, that was the reason for all those crazy astronaut training machines:
However, the syndrome was unknown in the first manned space flights – none of the 26 astronauts who flew in the 16 Mercury and Gemini space trips experienced disorientation or motion sickness.

However, this changed with the Apollo missions, where there were 11 incidents of inflight motion sickness, ranging from mild to severe, in as many flight missions. This was attributed to the increased opportunity for movement by the crewmen within the relatively large volume of the combined Apollo command and lunar modules compared to the very confined crew compartments of the Mercury and Gemini spacecraft.

The problem got even worse with Skylab, where 5 of 9 astronauts experienced symptoms of motion sickness during the initial days of the flight, 2of them severe including vomiting.
Anti-motion sickness drugs used by the Skylab 3 and 4 crewmen were not completely effective in ameliorating symptoms.

Interestingly, these symptoms of motion sickness were temporary, and all resolved within a few days.
In fact, in-flight experiments conducted on or after day 8 showed that all crewmen had adapted to weightlessness, and did not experience any adverse symptoms in a spinning chair (30 rpm) sufficient to cause vertigo on Earth.
We have already blogged about how the vestibular system detects head movements.
Maintaining an awareness of the relative location of our body parts requires the precise integration of visual, vestibular, and proprioceptive (touch, pressure, and stretch receptors in our skin, muscles, and joints) sensory inputs.
If your head moves in space, your eyes see the movement, but in zero-gravity the vestibular otolith doesn’t move and there is no proprioceptive input from the feet against the floor.
Space sickness is felt to be the result of “sensory conflict” or sensory mismatch.
It didn’t occur in the early Mercury and Gemini flights, because the astronauts spent the whole flight strapped into a seat inside a small capsule with limited opportunity for movement and minimal exposure to conflicting visual, motor, and vestibular sensory messages.
However, space sickness has affected >50% of astronauts since Apollo, beginning within the first hour of transition from Earth gravity to microgravity, and persisting for 2 to 3 days. It’s so predictable that no space walks were scheduled for the first 3-days of any Space Shuttle missions.
Recent research has suggested that virtual reality training can simulate specific effects of microgravity and may prove to be an effective countermeasure against space motion sickness through a process of habituation.

xx
Space Headache

Space flights can also trigger headaches. In a 2009 study 71% of astronauts reported headaches – occurring during launch, flight, activities outside the space station and landing. None had a history of recurrent headache on earth. There was little to no association with the main symptoms of space motion sickness, such as nausea, vomiting or vertigo.
Asked to describe the headache, the astronauts mostly said the symptoms were “exploding” or “heavy feeling.”
We know that blood volume gets redistributed to the brain and upper body when the astronaut floats in zero gravity.
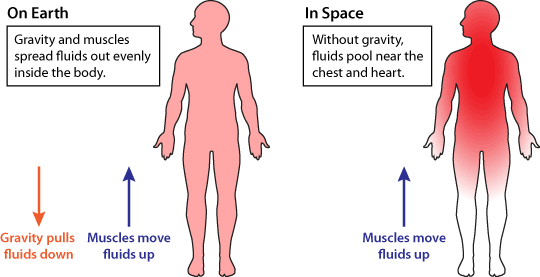
This shift of blood towards the brain causes a painful increase in pressure within the skull:
Click here to find out more.



























